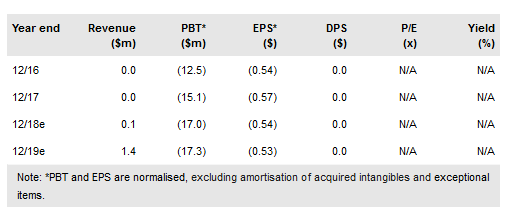
Volitionrx Ltd (NYSE:VNRX) has broad R&D program and expects to announce major milestones in the coming months, culminating with CE marking and launch of both colorectal cancer (CRC) triage and frontline screening tests in Europe, potentially in 2019. Other recent positive developments include a recommendation to reduce age to start CRC screening in the US and a new legislative initiative that could help increase reimbursement coverage in the US. Our valuation is $243m or $7.02 per share (vs $6.93 per share).
Start of commercial phase with research use sales
In May 2018, VolitionRx announced a global sales and distribution agreement with Active Motif that will distribute kits based on VolitionRx’s technology for research use. While VolitionRx focuses on cancer (CRC screening primarily), the Nu.QTM assays can be used in other conditions. This is not likely to lead VolitionRx to break even; however, the benefits of such business include a high-margin royalty stream, increasing awareness of the Nucleosomics technology within the research community and obtaining insights into various other applications for Nu.Q assays.
To read the entire report Please click on the pdf File Below: