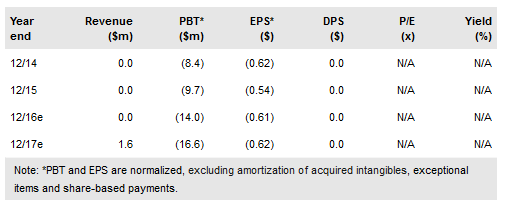
Volitionrx Ltd. (NYSE:VNRX) reported in January 2017 that it had received a CE mark for its Nu.Q™ triage test for colorectal cancer (CRC). The company is targeting commercialization of the test initially in EU member states that have established fecal immunochemical testing (FIT) programs (of which there are 14 in total), and the CE mark should enable launches in these territories as early as 2017.
Triage test can reduce colonoscopies by 25%
In October 2016 VolitionRx presented data at the European Society for Medical Oncology detailing the results from a 1,907-person prospective clinical trial of the Nu.Q™ triage test. The results demonstrated that when combined with a FIT, the protocol could maintain 96.6% of CRC diagnoses compared to current regimens, while reducing unnecessary colonoscopies by 25%. Additional results are to be released in February 2017 at the ENDO 2017 World Congress of GI Endoscopy.
To read the entire report Please click on the pdf File Below