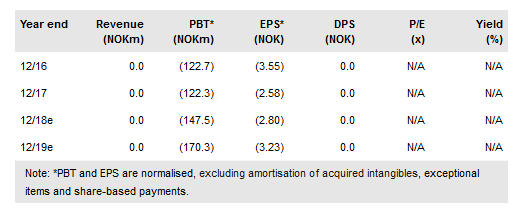
On 12 June, Targovax ASA (LON:0RISol) announced a strategic decision to move away from developing TG01, a neoantigen cancer vaccine for resected pancreatic cancer after new data with FOLFIRINOX set the bar for overall survival too high. With median overall survival of resected pancreatic cancer patients approaching five years, such a long clinical trial is beyond the capabilities of a relatively small biotech company, according to Targovax. We have removed pancreatic cancer from our valuation, which is now NOK1.31bn or NOK24.9/share (vs NOK33.8/share previously).
New FOLFIRINOX data at ASCO
The FOLFIRINOX regimen has been shown to be effective before, but has also demonstrated increased toxicity. Adjuvant chemotherapy with gemcitabine (and with capecitabine more recently) was considered to be standard of care. The new Phase III data presented at ASCO suggest an almost two-year improvement in overall survival in the FOLFIRINOX arm compared to gemcitabine. Even though more patients receiving FOLFIRINOX experienced side effects, severe side effects were prevalent in both groups. During the Q&A call Targovax management shared its impression from the interactions with various stakeholders at the ASCO meeting last week that FOLFIRINOX is likely to become standard of care in this indication. Given that Targovax recently presented positive data with TG01 from the second pancreatic cancer cohort in the Phase I/II trial, we believe the decision was not an easy one to make. However, the company was about to start preparation for the next Phase II, hence the “go/no-go” decision had to be made swiftly.
To read the entire report Please click on the pdf File Below: