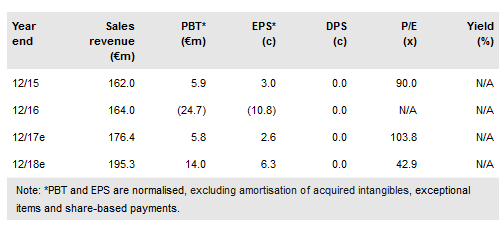
Pharma Mar (MC:PHMR) is approaching two key milestones in H217: an approval decision for Aplidin for multiple myeloma in Europe; and Phase III results for lurbinectedin in ovarian cancer.
The Chugai licence deal for lurbinectedin in Japan has strengthened the company’s financial position (pro forma net debt €32m) and seen it put increased emphasis on its preferred strategy to either self-commercialise or co-promote lurbinectedin in the U.S. Separately, a U.S. manufacturing patent granted last year has extended IP protection for lurbinectedin until at least December 2032. These developments have prompted us to adopt co-promotion in the U.S. in our base case valuation scenario and to extend our rNPV model to 2035 vs 2030 previously. Our base case valuation has increased by 29% to €1.29bn (vs €1.01bn), or €5.79/share (vs €4.55/share).
Lurbinectedin ovarian Phase III results in H217
Results from the 443-patient Phase III trial of lurbinectedin in platinum-resistant ovarian cancer are expected in H217. In an earlier Phase IIb trial, progression-free survival (PFS) was much greater for lurbinectedin than for topotecan (5.7 vs 1.7 months), which bodes well for the trial outcome.
To read the entire report Please click on the pdf File Below