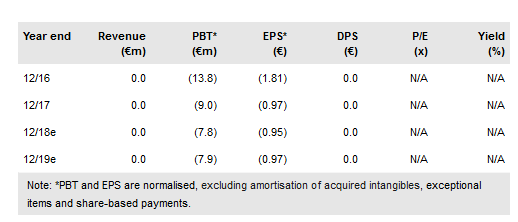
The highlight of Probiodrug's (AS:PDB) FY17 results presentation was the rather detailed introduction of the Phase IIb development programme for the lead asset PQ912, a small molecule inhibitor of glutaminyl cyclase (QC) for Alzheimer’s disease (AD) patients. Two Phase IIb trials (in Europe and the US) are designed to gather the amount of data that, if sufficiently positive, could allow for accelerated or conditional regulatory approval. The first Phase IIb study in Europe is expected to start by end 2018 and Probiodrug is exploring all options for funding sources. After several modest changes to our model, our updated valuation is slightly higher at €513m, €62.4/sh.
European/US Phase IIb studies could lead to market
The preliminary design for the European Phase IIb trial SAPHIR 2 have been presented. The design of the Phase IIb US study should be substantially similar in regard to patent population and titration, but will likely include additional readouts and treatment period will be 18 months including an interim analysis. The primary endpoint is PQ912’s effect on cognitive function measured by the neuropsychological test battery (NTB). NTB was one of the exploratory endpoints in the Phase IIa trial with PQ912 and showed initial signs of positive changes in cognitive function of AD patients after a short, three-month treatment period. AD patients will be treated for about a year on average in the upcoming European trial. If the data are sufficiently positive from both the US and European trials, Probiodrug expects to initiate a discussion with the regulatory authorities for accelerated or conditional approval (we assume in 2023).
To read the entire report Please click on the pdf File Below: