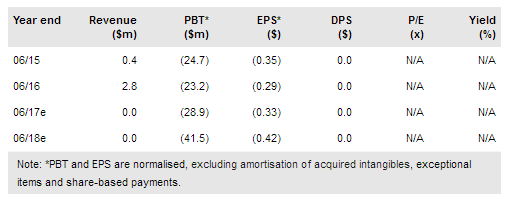
Pluristem (NASDAQ:PSTI) reported data from its pilot study of PLX-R18 for the treatment of acute radiation syndrome (ARS). The study included 48 non-human primates (NHP) who were dosed with 4m, 10m, and 20m cells per kg and showed an improvement in survival to 83%, 86% and 67%, respectively, from 50% in the control arm, although the study was not powered to significance. The company will need to perform a pivotal primate study and a human safety study for approval, which we expect in 2019-20
ARS data compares favourably to Neupogen
Neupogen is currently stockpiled for hematopoietic recovery following ARS. It was approved for this in 2015 on the basis of a 46-subject NHP study that showed an improvement of survival to 79% from 59% in control (p= 0.023). Also Pluristem stated that PLX-R18 supports the recovery of multiple cell lineages (Neupogen only supports white blood cells), although these data in NHP have not been released.
Benign safety profile could enable simple dosing
Pluristem stated that it did not observe any safety issues with PLX-R18 in healthy primates. This means that the treatment could potentially be administered without concern to a person’s radiation exposure as it would not pose a risk to healthy individuals, compared to Neupogen, which requires frequent blood testing to avoid overdose risk. The ability to indiscriminately dose individuals is a distinct advantage to PLX-R18 in the event of an emergency radiation exposure.
To read the entire report please click on the pdf file below: