Myeloma filed, breast results by year end
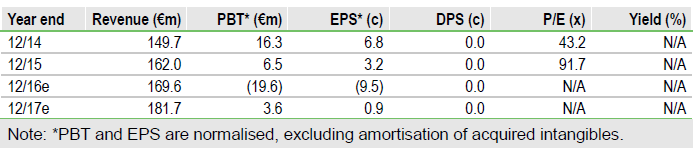
Pharma Mar SAU (MC:PHMR) has delivered impressive progress in 2016, including filing for Aplidin approval for myeloma in Europe, and the launch of small cell lung cancer (PM1183) and lymphoma (Aplidin) pivotal trials. News likely before year-end includes breast cancer Phase II results and full recruitment in the PM1183 ovarian Phase III. H116 accounts released in July showed Yondelis royalties ahead of our forecasts after approvals in US and Japan in Q415; however we were too optimistic about the boost FDA approval would give to European sales, so we have trimmed sales forecasts for Europe. This cuts our valuation to €1.02bn (€4.58/share) from €1.09bn (€4.91/share).
Aplidin filed for myeloma in Europe
PharmaMar has filed for approval for Aplidin in myeloma in Europe (which could lead to approval in H217), earning it a A$4m milestone from partner Chugai. In Phase III Aplidin reduced the risk of myeloma progression or death by 35%. A pivotal trial in the rare angioimmunoblastic T-cell lymphoma, which was initiated in June, could support an application for US approval, potentially around 2020.
To read the entire report Please click on the pdf File Below