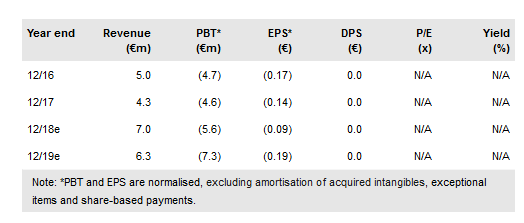
The €13m (gross) private placement announced on 30 October 2018 will support Oryzon Genomics' (MC:ORY) expanding R&D programme with a total of five clinical trials now underway. Two new Phase IIa trials with iadademstat (formerly ORY-1001) are starting in acute myeloid leukaemia (AML) (ALICE trial) and small cell lung cancer (SCLC) (CLEPSIDRA trial). Results from another two Phase IIa studies with vafidemstat (formerly ORY-2001) in Alzheimer’s disease (AD) patients (ETHERAL) and multiple sclerosis (MS) patients (SATEEN) are due to report results in H219. Before that, interim results from the innovative design ‘basket trial’ with vafidemstat in aggression control in various neuropsychiatric conditions are due in H119. Our valuation is €342m or €8.7 per share (vs €328m or €9.6 per share).
REIMAGINE trial to test holistic vafidemstat effects
Oryzon successfully initiated a further Phase IIa trial with vafidemstat, a dual LSD1/MAOB inhibitor, in aggression. The innovative trial design (basket trial) has been borrowed from drug development in oncology and will include patients with Lewy body dementia, AD, autism spectrum disorder, borderline personality disorder and attention deficit hyperactivity disorder. Aggression is often seen in a variety of neurodegenerative and neuropsychiatric conditions and vafidemstat has demonstrated holistic effects in this regard in preclinical models (more detailed overview in our recent outlook report). Oryzon hopes that testing it in different conditions will allow it to pin down a more precise psychiatric setting where the drug could be useful. Preliminary results are expected in H119.
To read the entire report Please click on the pdf File Below..