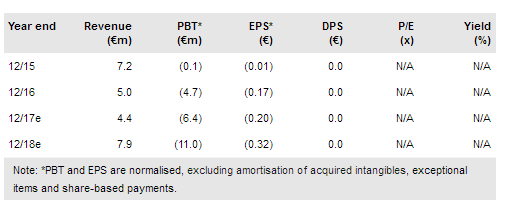
Roche’s decision to discontinue the development of ORY-1001 and return the rights to Oryzon Genomics SA (MC:ORY) was major recent news. Roche cited portfolio reprioritisation as the reason and that the decision was not driven by data. While this is a setback in business development for Oryzon, in our view, ORY-1001’s potential has not been compromised and the company indicated that it will continue developing ORY-1001 for both current indications, acute leukaemia and SCLC, before seeking a new partner for late-stage development.
On a more positive note, Oryzon reported positive final Phase I data for ORY-2001, which is now ready for further development in several neurological indications. Our valuation is €295m (vs €312m previously) or €8.6/share.
Next steps with ORY-1001
The handover process may take several months and during this period Roche will also finalise a dose-finding Phase I study in small cell lung cancer (SCLC) and hand the data over to Oryzon, which is now free to initiate planning for further clinical activities, although it will likely take some time to present updated ORY-1001 development plans.
Oryzon also mentioned that around the time ORY-1001 was out-licensed to Roche in April 2014, it was contacted by several other companies interested in epigenetic programmes in oncology. In our view, this suggests that the company could potentially replace Roche with another partner interested in epigenetics and LSD1 inhibition.
To read the entire report please click on the pdf file below: