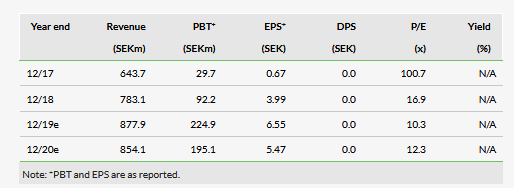
Orexo AB (ST:ORX)’s Q119 results reinforced its continuing profitability with total revenues rising 25% y-o-y to SEK174.3m. Zubsolv led the revenue charge comprising 93% of total revenues and growing 23% y-o-y despite the seasonally weak first quarter of the opioid use dependency market. The effect on US Zubsolv (which increased in price by 4% from January 2019) from four Suboxone film generic entrants is so far minor.
No real surprises in Q119
Growth of 23.3% in US Q119 Zubsolv revenues to SEK161.7m (SEK131.1m in Q118) continues to dominate Orexo’s investment case with a growing product despite an increasingly genericised market. Cost control continues to be a focus, with Orexo’s gross margin rising to 85% from 65% in Q118. This helped Q119 profit after tax rise to SEK14.1m compared to SEK25.9m in Q118, when traditionally Q1 is a loss-making quarter. This Q1 result was despite R&D investment in the pipeline and SEK49m in IP litigation costs. Orexo’s cash balance increased to SEK647.4m (SEK437.5m at the end of Q118). With litigation costs now expected to have a minor role for the rest of this year, the use of Orexo’s growing cash balance in M&A and in-licensing transactions will play an important role in diversifying the company’s future. The impact of the four generic Suboxone entrants on Zubsolv has been thankfully minor, but the changing market share dynamics will need to be closely watched. Ex-US Zubsolv partnerships in 2019 should be expected.
Litigation costs to decline in an appeal process
The earlier decision taken by the US District Court regarding patent ‘996 is unchanged. The court’s recent decision held that Actavis (NYSE:AGN) does not infringe patent ‘996 for its generic versions of Subutex and Suboxone, and we did not include any award for damages in our model. We have increased our FY19 total operational cost estimate mainly driven by a one-off SG&A expense to SEK538m, which remains close to Orexo’s c SEK500m guidance. Irrespective of declining IP legal costs for the remainder of FY19, Orexo’s ‘330 and ‘996 patents remain in force, ensuring US Zubsolv exclusivity until 2032.
Valuation: A number of moving parts
Our valuation takes into account a number of changes in the quarter and moves from SEK3.42bn or SEK97.7 per share to SEK3.52bn or SEK98.62 per share. Foreign exchange rates carried forward largely cancelled out the increased FY19e administrative expenses, while the reduction in FY19e CoGS from SEK115m to SEK112m and our introduction of a flat total market between Q4 and Q1 for future years resulted in the c 1% increase in our valuation.
Business description
Orexo is a Swedish speciality pharma company, with expertise in drug delivery/reformulation technologies (in particular, sublingual formulations) and a US commercial infrastructure for its opioid dependence therapy, Zubsolv (marketed by Orexo in the US and being out-licensed to partners ex-US). It also has three other clinical assets including OX124, which has reported positive Phase I results.