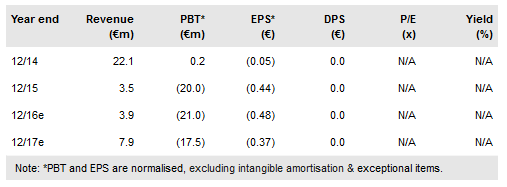
The major near-term catalyst for Onxeo (PA:C4X) will likely be the Livatag Phase III data in second-line liver cancer, which is expected around mid-2017. During 2017 we also expect new asset AsiDNA to advance into clinical testing, and progress with Beleodaq (belinostat), which could start the regulatory required Phase III trial. Onxeo is increasingly focused on exploring further opportunities for these assets, and has a number of preclinical collaborations ongoing. While data from these early-stage studies are not likely to be a major share price driver, in the longer term, these efforts could help to maximise value. Onxeo recently raised €12.5m to help fund these programmes. Our updated valuation is €350m.
Late-stage assets the main near-term drivers
The Phase III data for Livatag in second-line liver cancer are likely to be a major value inflection point for Onxeo. With the study almost fully recruited, Onxeo expects data from this event-driven trial to become available around mid-2017. We also expect the Phase III belinostat/CHOP chemotherapy combination trial in first-line peripheral T-cell lymphoma (PTCL) to start in coming months, along with initiation of the Phase I study of AsiDNA via iv administration. Our forecasts suggest that within Onxeo’s clinical pipeline, AsiDNA has the largest sales potential.
To read the entire report Please click on the pdf File Below