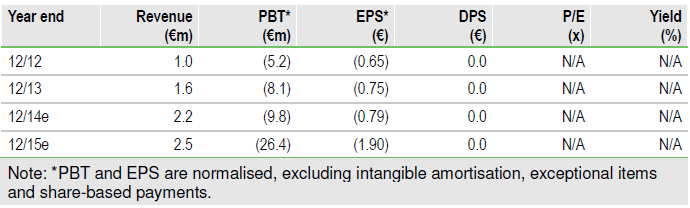
Capital raise allows execution of strategy
The successful €28.1m capital increase in March should allow Nanobiotix (PARIS:NANOB) to execute on its refined NBTXR3 strategy aiming for first CE mark approval in soft tissue sarcoma (STS) in 2016. Our valuation has increased to €315m with incorporation of the additional funds (from €282m). The next key catalyst will be presentation of STS data from the pilot trial at the highprofile ASCO conference in June.
€28.1m capital raise
Nanobiotix raised €28.1m gross proceeds in March from the issue of 2.65m new shares through a pre-emptive rights issue at a price of €10.60. The rights issue was 1.8x covered. We estimate the pivotal STS trial could cost around €15m, hence this cash could be sufficient to fund STS through to CE mark approval in 2016 and first sales, assuming STS development is prioritised over other potential indications.
Additional funds will allow execution of STS strategy
Nanobiotix recently refined its strategy to realise value from NBTXR3 with plans to conduct a pivotal STS trial. This could start by year end 2014, potentially allowing for first approval (CE mark as a medical device) in 2016 and first self-pay sales in late 2016. We continue to expect full NBTXR3 launch supported by reimbursement in other indications around 12-18 months later, for which additional funds could be needed, in the absence of other additional income, to complete development.
NBTXR3 STS data to be presented at ASCO
Data from the ongoing pilot STS trial will be presented at the ASCO (American Society of Clinical Oncology) clinical meeting, from 31 May to 3 June in Chicago. This is a high-profile medical meeting that should help raise awareness of NBTXR3 in the US. During 2014 we also expect partner PharmaEngine to start clinical development in Asia, we could get initial data from the ongoing pilot head and neck cancer trial, the pivotal STS trial could start, and Nanobiotix could secure a partner for NBTXR3 in the US, although we have limited visibility on the latter.
To Read the Entire Report Please Click on the pdf File Below