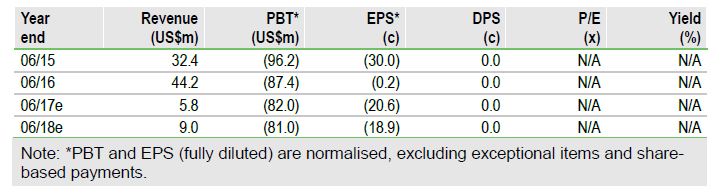
Mesoblast Ltd (NASDAQ:MESO) has raised US$40m (gross) through a placement of 26.25m shares at A$2.00/share. The cash enables continued funding of the key MPC-150-IM Phase III heart failure (HF) study where an interim futility analysis is underway. We expect Q4 data from the Phase III in paediatric graft vs host disease (GvHD); Mallinckrodt (NYSE:MNK) has an option on partnering GvHD. Other projects can also be funded. Also in H2, top-line data from the NIH-funded Phase IIb study in end-stage HF with an LVAD may be reported. The indicative value has been increased to A$1.72bn from A$1.67bn to reflect reduced potential FY18 cash needs. The indicative value per share drops to A$4.02 from A$4.16 due to the increased dilution.
GvHD and back pain covered by Mallinckrodt option
Mallinckrodt Pharmaceuticals has an exclusive negotiation period, which runs until September, covering Mesoblast’s Phase III chronic low back pain (CLBP) and GvHD programmes. A paediatric GvHD Phase III is expected to complete enrolment in mid-2017 and read out top-line data in Q417. The CLBP Phase III is on track to complete recruitment in H217. Mesoblast recently reported that improvements in pain and function after a single intra-disc injection of 6m MPCs in Phase II were maintained for at least 36 months in 84% of responders.
To read the entire report Please click on the pdf File Below