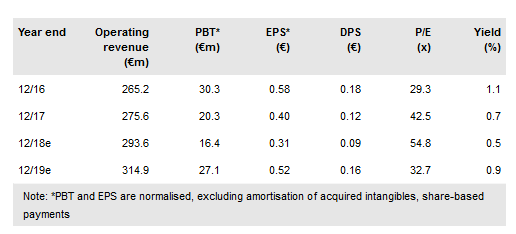
Laboratorios Farmaceuticos ROVI (MC:ROVI) has announced its pivotal Phase III PRISMA-3 trial continues on track and it expects to file an NDA with the FDA in the second half of 2019. The pre-specified DORIA interim analysis on the pivotal PRISMA-3 study has demonstrated that the study sample size (number of patients enrolled) does not need to be increased. We forecast efficacy data from the Phase III PRISMA-3 trial will be available in Q219 with a potential launch in 2021 in both the US and EU. We forecast peak sales opportunities of US$411m (US and EU) at a high gross margin, which has the potential to be highly value enhancing to ROVI’s long-term profit growth. We value ROVI at €1.16bn or €23.3/share.
The PRISMA-3 study is a multicentre, randomised, double-blind, placebo-controlled Phase III trial designed to evaluate the efficacy and safety of monthly intramuscular injections of DORIA in patients with acute exacerbation of schizophrenia. Post market close yesterday, ROVI announced the unblinded interim analysis of the PRISMA-3 clinical trial for DORIA. The intention of the interim analysis was to see whether the sample size required for final analysis may need to be increased. The independent data monitoring committee has recommended the trial continue with current number of randomised patients. This means the potential filing date for the US NDA is likely to be in H219.
To read the entire report Please click on the pdf File Below: