Infinity Pharmaceuticals Inc. (NASDAQ:INFI) reported a loss of 18 cents in the second quarter of 2019, wider than a loss of 12 cents in the year-ago quarter. The loss was also wider than the Zacks Consensus Estimate of a loss of 15 cents.
The company recognized $0.26 million as royalty revenues, lagging the Zacks Consensus Estimate of $2 million. The company did not record any revenues in the year-ago quarter.
Infinity recognized the $30-million gross proceeds from the Copiktra royalty monetization as a liability on the balance sheet in accordance with accounting guidance for royalty monetization. The company is amortizing the liability to non-cash interest expenses every quarter.
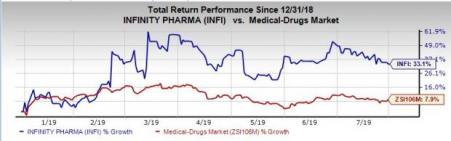
Shares of Infinity have soared 33.1% year to date compared with the industry’s growth of 7.9%.
2019 Outlook
The company expects a net loss for 2019 to be $40-$50 million.
Infinity expects its existing cash, cash equivalents and available-for-sale securities to be adequate to satisfy its capital needs in the second half of 2020.
Pipeline Updates
The company will initiate MARIO-3, a phase II study of novel triple combination front-line therapy, in clinical collaboration with Roche/Genentech (OTC:RHHBY) . Roche will supply Tecentriq (atezolizumab) to Infinity for use in MARIO-3. The study will evaluate the company’s lead immuno-oncology candidate, IPI-549, in combination with Tecentriq and Abraxane (nab-paclitaxel) in front-line, triple-negative breast cancer (TNBC), and IPI-549 in combination with Tecentriq and Avastin (bevacizumab) in front-line renal cell cancer (RCC). The study is anticipated to initiate in the third quarter of 2019.
Infinity initiated a global, randomized phase II study in collaboration with Bristol-Myers Squibb (NYSE:BMY) , which will evaluate IPI-549 in combination with Opdivoin second-line,immuno-oncology (I/O) naïve patients with advanced urothelial cancer.
The company expects to complete enrollment in the second half of 2019 in the expanded combination cohorts in MARIO-1, an ongoing phase I/Ib study of IPI-549 as a monotherapy and in combination with Opdivo in approximately 225 patients with advanced solid tumors.
In the third quarter of 2019, the company will also initiate a study in collaboration with Arcus BioSceinecs Inc. (NYSE:RCUS) to evaluate IPI-549 combined with the latter’s adenosine inhibitor, AB928, and Doxil in patients with advanced TNBC.
Roche Holding (SIX:ROG) AG (RHHBY): Free Stock Analysis Report
Bristol-Myers Squibb Company (BMY): Free Stock Analysis Report
Infinity Pharmaceuticals, Inc. (INFI): Free Stock Analysis Report
Arcus Biosciences, Inc. (RCUS): Free Stock Analysis Report
Original post
Zacks Investment Research