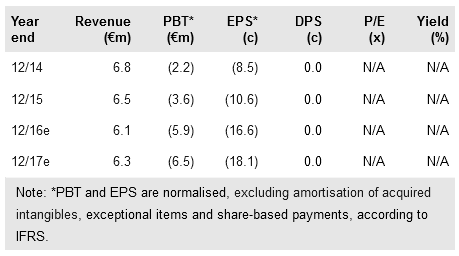
Hybrigenics (PA:ALHYG) reported €11.7m in cash at end 2015. This provides a runway to end 2017, funding a Phase II trial of inecalcitol in acute myeloid leukaemia (AML) in France and the US. Interim data from a Phase II study in chronic myeloid leukaemia (CML) are expected this year. Furthermore, the company has changed its accounting rules from French GAAP to IFRS; we have updated our model on this basis. This update, together with splitting inecalcitol revenues in chronic lymphocytic leukaemia (CLL) between the EU and US (reflecting different economics), increases our valuation to €137m from €120m.
Inecalcitol clinical trials continue to progress
Inecalcitol is being tested in a Phase II trial in CML in combination with Novartis's (NYSE:NVS) imatinib (Gleevec/Glivec), with interim data expected this year. Additionally, another Phase II trial in AML patients intolerant to current standard of care is slated to start dosing in the US in mid-2016. This study will recruit 110 patients and report overall survival data at the end of 2018. Inecalcitol is Phase III-ready in CLL after completing a Phase II trial; however, additional funding will be needed to conduct further studies in this indication as current cash is intended to fund the two former trials. We expect the company to secure the resources to continue this programme in two years’ time.
To read the entire report Please click on the pdf File Below