FDA agreement on the aldoxorubicin pivotal Phase III study in second-line soft tissue sarcoma (STS) paves the way for trial initiation in Q313 for CytRx (CYTR). Grant of the special protocol assessment (SPA) clarifies the US regulatory pathway, removing any doubt about the trial design, endpoints and statistical analysis. We now apply a 50% probability of success (previously 40%) to aldoxorubicin in second-line STS, which increases our rNPV to $138m or $4.50 per share. Looking forward, the next clinical milestones are aldoxorubicin Phase Ib data in advanced/metastatic solid tumours in Q213.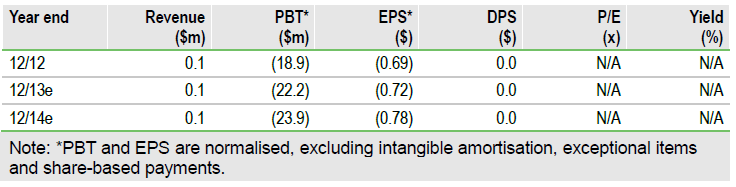
FDA agreement on aldoxorubicin Phase III trial…
CytRx and the FDA have agreed, on schedule, an SPA for the aldoxorubicin pivotal Phase III study in second-line STS. The SPA grant confirms that CytRx’s proposed design, endpoints and statistical analysis are acceptable for regulatory submission. In particular, we view FDA agreement on the primary endpoint (progression free survival, PFS) and comparator therapy (five approved chemotherapy regimens) as critical components of the SPA. While we assumed that PFS was the FDA’s preferred primary outcome measure, based on approval of GSK’s Votrient, we note that competing second-line STS drugs in Phase III (J&J’s Yondelis, Eisai’s Halaven) are using overall survival (OS). However, the FDA has confirmed that OS should be a key secondary endpoint in the aldoxorubicin pivotal study.
…paves way for enrolment in Q313
The global, open-label comparative Phase III study will enrol c 400 patients with advanced STS (metastatic, locally advanced, or unresectable disease) who are relapsed/refractory to prior chemotherapy. Patients will be randomised (1:1) to aldoxorubicin or the investigator's choice of chemotherapy (Votrient, dacarbazine, gemcitabine + docetaxel, doxorubicin or ifosfamide) with up to three comparator regimens per clinical site. FDA’s acceptance of multiple comparators (but with constraints) acknowledges the lack of standardised second-line therapy and should, in our view, ensure rapid patient recruitment through c 80 clinical sites. While CytRx has not disclosed the PFS benefit the trial is powered to show, we assume it exceeds the 3-month improvement seen with Votrient. Finally, we believe the trial is likely to include an interim efficacy/futility analysis based on PFS.
Valuation: rNPV rises to $138m
Our rNPV valuation rises to $138m ($4.50 per share) from $120m ($4.00 per share) following the SPA grant. We now apply a 50% probability of success to aldoxorubicin in second-line STS (previously 40%), which could rise to 60% following positive Phase IIb data in front-line STS (late-Q313).
To Read the Entire Report Please Click on the pdf File Below.
- English (UK)
- English (India)
- English (Canada)
- English (Australia)
- English (South Africa)
- English (Philippines)
- English (Nigeria)
- Deutsch
- Español (España)
- Español (México)
- Français
- Italiano
- Nederlands
- Português (Portugal)
- Polski
- Português (Brasil)
- Русский
- Türkçe
- العربية
- Ελληνικά
- Svenska
- Suomi
- עברית
- 日本語
- 한국어
- 简体中文
- 繁體中文
- Bahasa Indonesia
- Bahasa Melayu
- ไทย
- Tiếng Việt
- हिंदी
CytRx FDA Nod For Aldoxorubicin Phase III Trial
Published 04/25/2013, 08:26 AM
Updated 07/09/2023, 06:31 AM
CytRx FDA Nod For Aldoxorubicin Phase III Trial
Latest comments
Loading next article…
Install Our App
Risk Disclosure: Trading in financial instruments and/or cryptocurrencies involves high risks including the risk of losing some, or all, of your investment amount, and may not be suitable for all investors. Prices of cryptocurrencies are extremely volatile and may be affected by external factors such as financial, regulatory or political events. Trading on margin increases the financial risks.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
© 2007-2024 - Fusion Media Limited. All Rights Reserved.