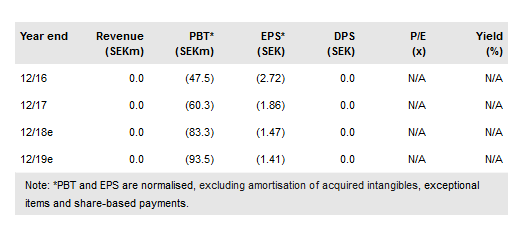
Cantargia AB (ST:CANTA) continues to progress its main R&D programme, a Phase I/IIa CANFOUR trial with its lead drug candidate, which was given its official generic name nidanilimab (CAN04 previously) in June. Cantargia reported no concerning side effects from the first 15 patients in the Phase I part of the CANFOUR trial. As the maximum tolerated dose has not been reached, this part will enrol several more patients, although Phase IIa should still start in Q418, as planned. Our valuation is slightly higher at SEK1.79bn or SEK27.1/share.
Phase IIa likely to start in Q418 as planned
Nidanilimab is a fully humanised, dual-action IgG1 antibody that targets IL1RAP and also has an antibody-dependent cellular cytotoxicity (ADCC) effect. In August 2018, Cantargia reported progress with its lead programme Phase I/IIa CANFOUR trial which tested nidanilimab in patients with various solid cancers (non-small cell lung cancer, pancreatic cancer, colorectal cancer or triple negative breast). The announcement focused on the Phase I part of the trial, which is a typical 3x3 dose escalation used to establish safety and the recommended dose for Phase IIa. In total 15 patients have been enrolled and in general, nidanilimab was well tolerated. As of the announcement date, the maximum tolerated dose has not been reached. Therefore, Cantargia will enrol several more patients before continuing to Phase IIa in Q418, as expected.
To read the entire report Please click on the pdf File Below: