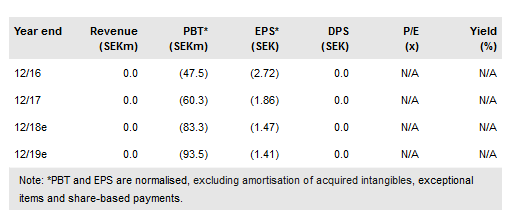
On 20 October 2018, Cantargia (ST:CANTA) presented interim data from part I of its Phase I/IIa CANFOUR trial with nidanilimab (IL1RAP antibody) at the ESMO congress in Munich, Germany, demonstrating a good safety/tolerability profile so far. The maximum tolerated dose has not been reached and so a final dose (10mg/kg) cohort is being recruited. The Phase IIa part of the study is expected to start in Q418 as planned. Meanwhile, on 25 September Cantargia’s shares were up-listed to the Nasdaq Stockholm main market, which will expose the company to a wider investment community. Our valuation is virtually unchanged at SEK1.80bn or SEK27.2/share with the success probability rate subject to revision once the final Phase I data are published in Q418.
Nidanilimab well tolerated so far
Phase I of the CANFOUR trial is testing nidanilimab in patients with solid cancers: non-small cell lung (NSCL), pancreatic, colorectal or triple negative breast cancers. Phase I was designed to establish the recommended dose for Phase IIa and to assess initial safety and tolerability (Exhibit 1). The data reported at ESMO were gathered from 16 patients. The patients received weekly infusions of between 1mg/kg and 6mg/kg and, in general, tolerated nidanilimab well with no treatment discontinuations or grade 4 or 5 adverse events. Infusion-related reactions were the most common adverse event (44%), as is often seen with other biologicals. The safety profile looks beneficial so far, in our view. Data from the higher dose cohort are expected in Q418, which is when the recommended dose will be selected.
To read the entire report Please click on the pdf File Below..