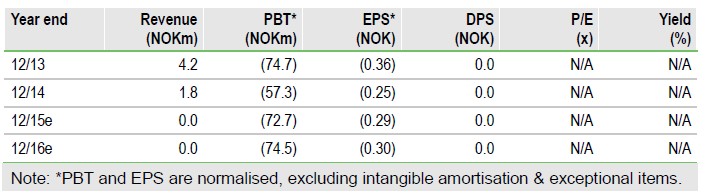
Prepping for the future; REDUC read-out in H215
Following encouraging interim data from the Phase II REDUC trial of Vacc- 4x with romidepsin, Bionor Pharma (OSLO:BIONOR) has set a number of initiatives in motion to fully capitalise on the potential opportunity that this ‘Kick and Kill’ approach could offer in HIV. This includes planning next development steps by convening an expert panel, securing future romidepsin supply and exploring potential financing options. These endeavours should ensure that Bionor will be well-placed to swiftly advance Vacc-4x, if further REDUC data expected in H215 are positive. Our valuation is NOK1bn.
Top-line REDUC data expected during H215
Bionor anticipates reporting top-line data from the ongoing Phase II REDUC trial (Vacc-4x in combination with romidepsin) during H215. This will be based on the 20 patients (who are well controlled on conventional antiretroviral therapy, ART, with low viral loads) recruited in the trial, and will likely include HIV viral load data (a measure of HIV RNA in the bloodstream and an indicator of disease control), which are measured regularly during the trial, in addition to viral rebound post a 16-week interruption of current ART. As a reminder, interim data from nine patients reported undetectable viral load in seven patients, with other measures suggestive that Vacc-4x with romidepsin could both reactivate and then destroy latent HIV reservoirs. The combination was safe and well-tolerated.
To Read the Entire Report Please Click on the pdf File Below