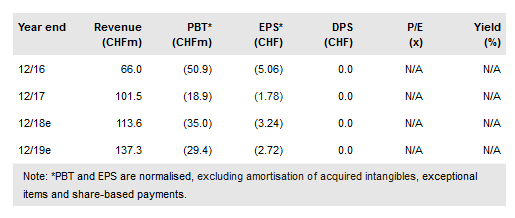
Basilea Pharmaceutica AG (SIX:BSLN) has successfully brought two anti-infective drugs to the market: Cresemba (severe mould infections) and Zevtera (bacterial infections). With the commercialisation of both assets largely in the hands of partners and Zevtera’s Phase III US clinical programme underway, we turn our focus to the next pillar of growth, the oncology portfolio. The recent deal with ArQule (in-licensing of Phase II product, derazantinib) means Basilea now has three diversified, early/mid-stage clinical assets targeting cancer resistance in its portfolio offering. We value Basilea at CHF119/share.
Derazantinib shoring up the oncology pipeline
In April 2018, Basilea announced the in-licensing of the worldwide (ex-Greater China) rights to ArQule’s derazantinib, a tyrosine kinase inhibitor (pan FGFR) with potential utility across a range of cancers that molecular test positive for FGFR genetic aberrations. Derazantinib’s most advanced indication is intrahepatic cholangiocarcinoma (iCCA, bile duct cancer). Interim data from the Phase II registration trial in iCCA are anticipated in H119 and Basilea plans to start Phase II development in FGFR-driven solid cancer types in H219.
To read the entire report Please click on the pdf File Below: