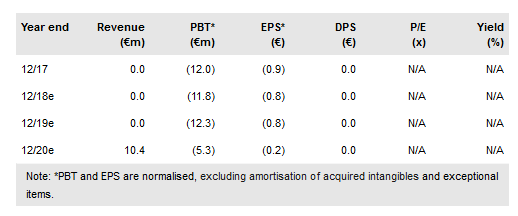
Asit Biotech SA (BR:ASIT) is an allergy immunotherapy (AIT) discovery and development company leveraging its proprietary ASIT+ platform. The lead product, gp-ASIT+ for grass pollen allergy, has already been tested in a Phase III trial. Although the study missed the predefined percentage difference in primary efficacy endpoint between placebo and the treatment group, a statistically significant reduction in allergy severity was demonstrated in the study. Having learnt the lessons and been encouraged by the opinion of the German regulatory authority, ASIT is now recharging for a confirmatory Phase III trial. Our valuation is €119.6m or €7.3/share.
Preparing confirmatory Phase III with gp-ASIT+
In February 2017, ASIT presented the results of BTT009, its most advanced multi-centre Phase III clinical study (n=554) evaluating the clinical efficacy of gp-ASIT+. Depending on the analyses used, the improvement in defined clinical score reached statistical significance with 15% (at peak allergy season) to 21% (over entire pollen season) reduction compared to placebo. However, the study missed the predefined average 20% difference in the scores between placebo and the treatment group over the peak season (primary endpoint) due to several confounding factors, including a weak pollen season. ASIT received scientific advice from the German regulator, the Paul-Ehrlich Institute, and is now preparing for the redesigned confirmatory Phase III study, planned to start in late 2018.
To read the entire report Please click on the pdf File Below: