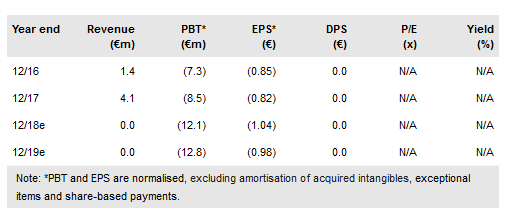
Crossject (PA:ALCJ) recently reported FY17 results and that it remains on track for regulatory filings for several programmes in 2019, including Zeneo Sumatriptan, Midazolam, Epinephrine/Adrenaline, Hydrocortisone, and Naloxone with Terbutaline and Methotrexate in 2020. A key upcoming catalyst would be a US licensing agreement for Zeneo Sumatriptan for the treatment of acute migraine, which could come in H118.
Manufacturing capabilities progressing
Crossject is continuing to improve its manufacturing capabilities, an important component of the company’s ability to meet its regulatory and commercial timelines. It has been able to produce ready-to-fill sterile tubes since the end of 2017 and the production line for the gas generators has been operational since the beginning of 2018.
To read the entire report Please click on the pdf File Below: