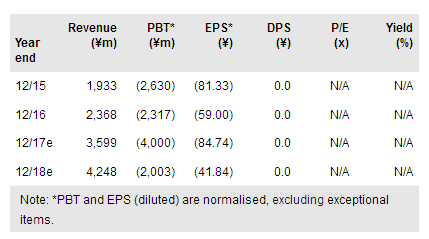
Symbiox Investment & Trading Co Ltd (BO:SYMB) has substantially increased the value of its Treakisym franchise through in-licencing novel bendamustine formulations from Eagle Pharmaceuticals Inc (NASDAQ:EGRX) for $12.5m. The new formulations bring a lower cost of goods and 10 years of additional patent protection to help in life cycle management of the Treakisym franchise. Sales of Treakisym following the approval of two new indications in 2016 have exceeded the company’s expectations, leading it to upgrade FY17 sales guidance. Interim data from the global Phase III rigosertib trial could become available during Q417. We value SymBio at ¥19.7bn (unchanged), with the enhanced Treakisym portfolio offsetting the suspension of IONSYS development.
Replicating Teva’s strategy to maintain market share
SymBio has in-licensed two liquid formulations of bendamustine hydrochloride (Treakisym) to complement its marketed lyophilised/dry powder product: a ready-to-dilute (RTD) formulation and a rapid infusion (RI) formulation that will cut drug infusion time from 60 minutes to 10 minutes. Patents on the RTD and RI formulations extend to 2031, whereas orphan exclusivity on Treakisym powder expires in October 2020. SymBio is aiming to replicate the strategy that has seen Teva transfer 97% of bendamustine HCl sales in the US to the RI formulation.
To read the entire report Please click on the pdf File Below: